
The requirement that a reversible reaction be at equilibrium is that:
- the concentrations on the two sides of the arrow be equal
- the velocity for the forward reaction equal that of the reverse reaction
- there will be as many molecules of the substances on one side of the arrow as there are molecules of the substances on the other side
- the moles of products will equal the moles of reactants

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
What is the relationship of air masses and the temperature of oceans?
Answers: 1

Chemistry, 22.06.2019 03:30
At a temperature of 393 k, the temperature of a sample of nitrogen is 1.07 atm what will the pressure be at a temperature of 478 k
Answers: 1

Chemistry, 22.06.2019 03:30
What is the number of moles of chemical units represented by 9.03x10^24? and how do i show work? (dumb it down )
Answers: 1

Chemistry, 22.06.2019 11:30
Aperfume bottle is dropped in the corner of a room. the odor of the perfume can be detected on the other side of the room. which statement best describes this observation?
Answers: 2
You know the right answer?
The requirement that a reversible reaction be at equilibrium is that:
- the concentrat...
- the concentrat...
Questions

Chemistry, 29.09.2020 03:01





Biology, 29.09.2020 03:01


Mathematics, 29.09.2020 03:01

Mathematics, 29.09.2020 03:01


English, 29.09.2020 03:01





Mathematics, 29.09.2020 03:01




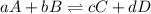
![K=\frac{[C]^c[D]^d}{[A]^a[B]^b}](/tpl/images/0438/0819/91a0a.png)


