Chemistry, 25.07.2019 18:10 tateandvioletAHS14AY
The combustion of titanium with oxygen produces titanium dioxide: ti (s) + o2 (g) → tio2 (s) when 2.060 g of titanium is combusted in a bomb calorimeter, the temperature of the calorimeter increases from 25.00°c to 91.60°c. in a separate experiment, the heat capacity of the calorimeter is measured to be 9.84 kj/k. the heat of reaction for the combustion of a mole of ti in this calorimeter is kj/mol.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:10
Which of the following elements would you expect to have the lowest ionization energy value? fluorine, lithium, neon, nitrogen
Answers: 2

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 11:50
If oil spills continue, all of the following should be expected except (2 points) death of aquatic life. polluted groundwater. decreased soil productivity. increased global temperatures.
Answers: 3

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2
You know the right answer?
The combustion of titanium with oxygen produces titanium dioxide: ti (s) + o2 (g) → tio2 (s) when 2...
Questions


Mathematics, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30



Mathematics, 06.11.2020 21:30

Social Studies, 06.11.2020 21:30


Health, 06.11.2020 21:30


Mathematics, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30



Arts, 06.11.2020 21:30



Arts, 06.11.2020 21:30

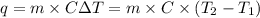
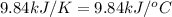
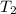 = final temperature =
= final temperature = 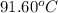
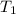 = initial temperature =
= initial temperature = 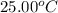
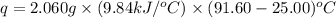
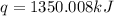
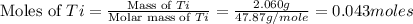
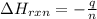
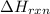 = enthalpy of reaction = ?
= enthalpy of reaction = ?