Chemistry, 30.07.2019 22:20 palcochran1313
Given the following balanced reaction between liquid ammonia and oxygen gas to produce nitrous oxide gas and water, how many grams of water, h2o, are produced from 317 grams of ammonia and excess oxygen? (to find the molar mass in the problem use the periodic table and round the mass to the hundreds place for calculation).
(a) 224 g
(b) 335 g
(c) 503 g

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:00
According to the tide table below what time of day will the highest tide occur?
Answers: 1


Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

Chemistry, 22.06.2019 13:30
In a ni-cd battery, a fully charged cell is composed of nickelic hydroxide. nickel is an element that has multiple oxidation states. assume the following proportions of the states: nickel charge proportions found 0 0.17 +2 0.3 +3 0.33 +4 0.5 (a) determine the mean of the nickel charge. enter the answer to 2 decimal places.(b) determine the cumulative distribution function of nickel charge.
Answers: 2
You know the right answer?
Given the following balanced reaction between liquid ammonia and oxygen gas to produce nitrous oxide...
Questions

Computers and Technology, 27.04.2021 14:40


Computers and Technology, 27.04.2021 14:40



Mathematics, 27.04.2021 14:40













Computers and Technology, 27.04.2021 14:40

Mathematics, 27.04.2021 14:40

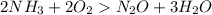
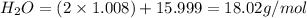
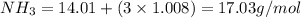
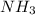 to moles
to moles 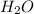 by using mole ratio of
by using mole ratio of 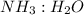 i.e., 2 : 3
i.e., 2 : 3
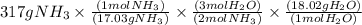
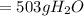 is formed.
is formed.
 is
is