
Chemistry, 27.08.2019 17:10 thevampsrock3
Certain parts of the world don't have enough fresh drinking water. one possible solution is to tow icebergs from antarctica and melt them as needed. if 6.10 ✕ 1011 kj of energy were needed to melt an iceberg, how much did the iceberg weigh? the enthalpy of fusion (melting or freezing) for ice is 6.01 kj/mol, and 1 metric ton = 1,000 kg. metric tons

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 12:50
Use the standard enthalpies of formation for the reactants and products to solve for the δhrxn for the following reaction. (the δhf of c2h4 is 52.26 kj/mol, co2 is -393.509 kj/mol, and h2o is -241.818 kj.) c2h4 (g) + 3o2(g) 2co2 (g) + 2h2o(g) δhrxn = the reaction is .
Answers: 3


Chemistry, 22.06.2019 02:20
Calculate the molarity of 48.0 ml of 6.00 m h2so4 diluted to 0.250 l .
Answers: 1

You know the right answer?
Certain parts of the world don't have enough fresh drinking water. one possible solution is to tow i...
Questions






Business, 26.03.2020 05:21




History, 26.03.2020 05:21


English, 26.03.2020 05:21





Biology, 26.03.2020 05:21



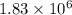 metric ton
metric ton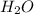 = 18 g
= 18 g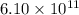 kJ of heat energy is needed to melt
kJ of heat energy is needed to melt 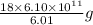 of ice or
of ice or  g of ice
g of ice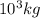 = 1 metric ton
= 1 metric ton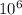 g = 1 metric ton ( 1 kg = 1000 g)
g = 1 metric ton ( 1 kg = 1000 g)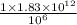 metric ton =
metric ton = 

