
Chemistry, 05.09.2019 23:10 portedon6626
Write qc for each of the following: (1) gaseous sulfur tetrafluoride reacts with liquid water to produce gaseous sulfur dioxide and hydrogen fluoride gas. (2) solid molybdenum(vi) oxide reacts with gaseous xenon difluoride to form liquid molybdenum(vi) fluoride, xenon gas, and oxygen gas.

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 13:00
These questions are based on the attached photo. the experiment is about burning magnesium metal with oxygen. 1. write the balanced chemical equation for the reaction you are performing. 2. calculate the mass of magnesium metal used in each trial. o trial 1: o trial 2: 3. calculate the actual yield of magnesium oxide for each trial. o trial 1: o trial 2: 4. magnesium is the limiting reactant in this experiment. calculate the theoretical yield of mgo for each trial. o trial 1: o trial 2: 5. determine the percent yield of mgo for your experiment for each trial. o trial 1: o trial 2: 6. determine the average percent yield of mgo for the two trials. your company currently uses a process with a similar cost of materials that has an average percent yield of 91 percent. if the average percent yield of this process is higher than that, this could save the company money. what is your recommendation to the company? support your recommendation using your data, calculations, and understanding of stoichiometry gathered from this lab.
Answers: 1

Chemistry, 22.06.2019 13:00
How many moles of sulfur dioxide are produced when 4.38 moles of oxygen completely react with iron (iv) sulfide
Answers: 2
You know the right answer?
Write qc for each of the following: (1) gaseous sulfur tetrafluoride reacts with liquid water to pro...
Questions

Mathematics, 31.10.2020 08:00

History, 31.10.2020 08:00

Mathematics, 31.10.2020 08:00


Mathematics, 31.10.2020 08:00

Mathematics, 31.10.2020 08:00



Mathematics, 31.10.2020 08:00

History, 31.10.2020 08:00


Mathematics, 31.10.2020 08:00



Chemistry, 31.10.2020 08:00


Mathematics, 31.10.2020 08:00

English, 31.10.2020 08:00

Mathematics, 31.10.2020 08:00

Chemistry, 31.10.2020 08:00

![Q_c=\frac{[SO_2][HF]^4}{[SF_4]}](/tpl/images/0223/9095/5d98b.png)
![Q_c=\frac{[O_2]^2[Xe]}{[XeF_2]}](/tpl/images/0223/9095/5b7c4.png)
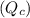 : It is defined as the measurement of the relative amounts of products and reactants present during a reaction at a particular time.
: It is defined as the measurement of the relative amounts of products and reactants present during a reaction at a particular time.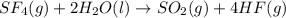
![2MoO_2(s)+XeF_2(g)\rightarrow 2MoF(l)+Xe(g)+2O_2(g)[/texIn this expression, only gaseous or aqueous states are includes and pure liquid or solid states are omitted. So, the expression for reaction quotient will be :[tex]Q_c=\frac{[O_2]^2[Xe]}{[XeF_2]}](/tpl/images/0223/9095/8e63c.png)


