
Chemistry, 06.09.2019 21:20 villafana36
The complete oxidation of one mole of a sugar produces carbon dioxide and water. 2000 kj of heat is transferred from the system to the surroundings. the rearrangement of bonds as 0.5 moles of the sugar are oxidized generates heat in an open test tube (101 j•l–1 pressure and 300 k temperature). what is the change in internal energy of the system (δu)? what is the change in enthalpy of the system(δh)?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 17:10
Increasing the substrate concentration in an enzymatic reaction could overcome which of the following? a) the need for a coenzymeb) allosteric inhibitionc) competitive inhibitiond) insufficient cofactors
Answers: 1

Chemistry, 23.06.2019 00:00
The empirical formula of a compound is ch2o and its mass is 120 amu/molecule, what is its formula?
Answers: 1

Chemistry, 23.06.2019 01:00
Aman applies a force of 500n to push a truck 100m down the street how much does he do?
Answers: 1
You know the right answer?
The complete oxidation of one mole of a sugar produces carbon dioxide and water. 2000 kj of heat is...
Questions

History, 03.07.2019 01:30


Chemistry, 03.07.2019 01:30

Biology, 03.07.2019 01:30


Mathematics, 03.07.2019 01:30

Mathematics, 03.07.2019 01:30

Mathematics, 03.07.2019 01:30

Mathematics, 03.07.2019 01:30


History, 03.07.2019 01:30

Mathematics, 03.07.2019 01:30

Biology, 03.07.2019 01:30






Mathematics, 03.07.2019 01:30

English, 03.07.2019 01:30





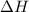 ) is -1000 kJ.
) is -1000 kJ.

