
Chemistry, 18.09.2019 23:10 lauraabosi
Three positive charges lie on the x axis: q1 = 1 × 10−8 c at x1 = 1 cm, q2 = 2 × 10−8 c at x2 = 2 cm, and q3 = 3 × 10−8 c at x3 = 3 cm. the potential energy of this arrangement, relative to the potential energy for infinite separation, is about:
1. 0j2. 0.16j3. 0.079 j4. 0.00085 j5. 0.0017j

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
How many liters of water vapor can be produced if 108 grams of methane gas (ch4) are combusted at 312 k and 0.98 atm? show all work. pls ! will mark as brainliest
Answers: 1

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 11:00
As air becomes more dense, (select all that apply) o. air weighs less o. gas molecules are closer together o. air is colder o. air weighs more o. gas molecules are further apart o. air is hotter
Answers: 3
You know the right answer?
Three positive charges lie on the x axis: q1 = 1 × 10−8 c at x1 = 1 cm, q2 = 2 × 10−8 c at x2 = 2 c...
Questions

English, 19.11.2019 18:31











English, 19.11.2019 18:31






Mathematics, 19.11.2019 18:31

History, 19.11.2019 18:31

Mathematics, 19.11.2019 18:31

![\frac{1}{4 \pi \epsilon_{o}}[\frac{q_{1}q_{2}}{r_{12}} + \frac{q_{2}q_{3}}{r_{23}} + \frac{q_{3}q_{1}}{r_{31}}]](/tpl/images/0241/1476/8c7e5.png)
 ,
,  , and
, and  .
. 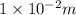 (as 1 cm = 0.01 m)
(as 1 cm = 0.01 m)![9 \times 10^{9} [\frac{1 \times 10^{-8} \times 2 \times 10^{-8}}{10^{-2}} + \frac{2 \times 10^{-8} \times 3 \times 10^{-8}}{10^{-2}} + \frac{3 \times 10^{-8} \times 1 \times 10^{-8}}{10^{-2}}]](/tpl/images/0241/1476/33bac.png)
![9 \times 10^{9} [2 + 6 + 1.5]](/tpl/images/0241/1476/ffcf8.png)
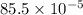 J
J


