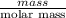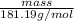Chemistry, 07.10.2019 20:00 mamasmontoya
You do an experiment in which you need 0.5 mole of tyrosine (c9h11no3) .
how many grams must you weigh out?
65 g
90.5 g
181 g
272 g

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 19:00
Convert the temperature of dry ice, –77 ∞c, into degrees fahrenheit and kelvin.
Answers: 2

Chemistry, 22.06.2019 20:00
Phosphoric acid is a triprotic acid ( =6.9×10−3 , =6.2×10−8 , and =4.8×10−13 ). to find the ph of a buffer composed of h2po−4(aq) and hpo2−4(aq) , which p value should be used in the henderson–hasselbalch equation? p k a1 = 2.16 p k a2 = 7.21 p k a3 = 12.32 calculate the ph of a buffer solution obtained by dissolving 18.0 18.0 g of kh2po4(s) kh 2 po 4 ( s ) and 33.0 33.0 g of na2hpo4(s) na 2 hpo 4 ( s ) in water and then diluting to 1.00 l.
Answers: 3
You know the right answer?
You do an experiment in which you need 0.5 mole of tyrosine (c9h11no3) .
how many grams...
how many grams...
Questions

Mathematics, 06.12.2019 19:31



English, 06.12.2019 19:31





Mathematics, 06.12.2019 19:31



Mathematics, 06.12.2019 19:31


Mathematics, 06.12.2019 19:31

History, 06.12.2019 19:31



History, 06.12.2019 19:31


Physics, 06.12.2019 19:31

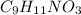 ) is 181.19 g/mol.
) is 181.19 g/mol.