
Chemistry, 13.10.2019 23:20 slowmotion
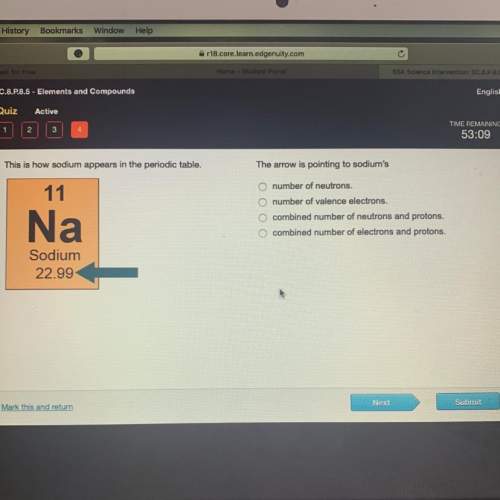
this is how sodium appears in the periodic table. the arrow pointing to sodium’s

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
Which step in naming unsaturated hydrocarbons is used for alkenes but not alkynes
Answers: 2



Chemistry, 22.06.2019 17:40
Areaction in which products can react to re-form reactants is
Answers: 1
You know the right answer?
this is how sodium appears in the periodic table. the arrow pointing to sodium’s
...
...
Questions

French, 21.03.2021 07:20

History, 21.03.2021 07:20

History, 21.03.2021 07:20


Engineering, 21.03.2021 07:20

Mathematics, 21.03.2021 07:30

English, 21.03.2021 07:30


Biology, 21.03.2021 07:30

Mathematics, 21.03.2021 07:30

Mathematics, 21.03.2021 07:30



Physics, 21.03.2021 07:30

Health, 21.03.2021 07:30

Arts, 21.03.2021 07:30

French, 21.03.2021 07:30





