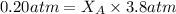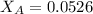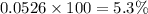Chemistry, 16.12.2019 17:31 kevinkingpin
Helium is mixed with oxygen gas for deep-sea divers. calculate the percent by volume of oxygen gas in the mixture if the diver has to submerge to a depth where the total pressure is 3.8 atm. the partial pressure of oxygen is maintained at 0.20 atm at this depth.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
1. baking powder is a 1: 1 molar mixture of cream of tartar (khc4h4o6) and baking soda (nahco3). a recipe calls for two teaspoons (a total of 8.0 grams) of cream of tartar. how much baking soda must be added for both materials to react completely?
Answers: 2

Chemistry, 22.06.2019 03:50
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 09:00
What type of energy do chemical bonds have? what type of energy is it converted to during chemical reactions? question 15 options: chemical bonds have kinetic energy, which is converted to potential energy during chemical reactions. chemical bonds have electric energy, which is converted to potential energy during chemical reactions. chemical bonds have heat energy, which is converted to kinetic energy during chemical reactions. chemical bonds have potential energy, which is converted to heat energy during chemical reactions.
Answers: 1

Chemistry, 22.06.2019 12:20
Adeuteron, 21h, is the nucleus of a hydrogen isotope and consists of one proton and one neutron. the plasma of deuterons in a nuclear fusion reactor must be heated to about 3.02×108 k . what is the rms speed of the deuterons? express your answer using two significant figures.
Answers: 1
You know the right answer?
Helium is mixed with oxygen gas for deep-sea divers. calculate the percent by volume of oxygen gas i...
Questions


Biology, 18.03.2021 01:00


Business, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

History, 18.03.2021 01:00




Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00


Business, 18.03.2021 01:00

Chemistry, 18.03.2021 01:00

History, 18.03.2021 01:00

Advanced Placement (AP), 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

Mathematics, 18.03.2021 01:00

World Languages, 18.03.2021 01:00

History, 18.03.2021 01:00

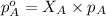
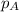 = total partial pressure of solution = 3.8 atm
= total partial pressure of solution = 3.8 atm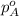 = partial pressure of oxygen = 0.20 atm
= partial pressure of oxygen = 0.20 atm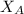 = mole fraction of oxygen = ?
= mole fraction of oxygen = ?