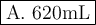Adiver exhales a bubble with a volume of 250 ml at a pressure of 2.4 atm and a temperature of 15 °c. what is the volume of the bubble when it reaches the surface where the pressure is 1.0 atm and the temperature is 27 °c? a. 630mlb. 110 mlc. 580 mld. 1100 mle. 100 ml

Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

You know the right answer?
Adiver exhales a bubble with a volume of 250 ml at a pressure of 2.4 atm and a temperature of 15 °c....
Questions


History, 26.07.2019 23:50






Mathematics, 26.07.2019 23:50



Biology, 26.07.2019 23:50




Health, 26.07.2019 23:50


Mathematics, 26.07.2019 23:50


Mathematics, 26.07.2019 23:50