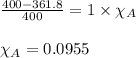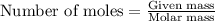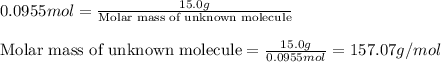Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
Which statement best describes the relationship between period and frequency of light waves? a) in wave b the period increases and the frequency decreases from wave a. b) in wave a the period increases and the frequency decreases from wave b. c) in wave b the period is shorter and the frequency is greater than in wave a. d) in wave a the period is shorter and the frequency is greater than in wave b.
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2

You know the right answer?
At 39.5 o C, the vapor pressure of pure acetone (MM = 58.08 g/mol) is 400.0 torr. If 15.0 grams of a...
Questions









Chemistry, 05.05.2020 16:21

Mathematics, 05.05.2020 16:21





Social Studies, 05.05.2020 16:21

Chemistry, 05.05.2020 16:21

Physics, 05.05.2020 16:21



Biology, 05.05.2020 16:21

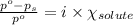
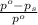 = relative lowering in vapor pressure
= relative lowering in vapor pressure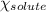 = mole fraction of solute = ?
= mole fraction of solute = ?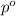 = vapor pressure of pure acetone = 400 torr
= vapor pressure of pure acetone = 400 torr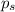 = vapor pressure of solution = 361.8 torr
= vapor pressure of solution = 361.8 torr