

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Identify the electromagnets with poles that are reversed from the electromagnet shown above
Answers: 3

Chemistry, 22.06.2019 10:30
Skills of homo sapiens were found an excavation. the skulls were preserved because the bodies were frozen. so, these fossils are (blank) fossils.the image shows the evolution of skulls beginning 2 to 3 million years ago. based on the image, modern human skulls(blank) ape skulls.
Answers: 1

Chemistry, 22.06.2019 11:00
Surface currents are caused by blank space . question 14 options: surface currents are caused by? differences in water temperature high salinity differences in density wind forces
Answers: 1

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3
You know the right answer?
Use the standard reaction enthalpies given below to determine ΔH°rxn for the following reactionP4(g)...
Questions

Geography, 27.01.2021 04:20



Mathematics, 27.01.2021 04:20


Spanish, 27.01.2021 04:20


Mathematics, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20




Biology, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20

English, 27.01.2021 04:20

Social Studies, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20

Mathematics, 27.01.2021 04:20

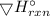 = -1835 KJ
= -1835 KJ

