
Chemistry, 28.02.2020 23:27 emobirdy1122
Consider the following multistep reaction: A+B→AB(slow) A+AB→A2B(fast)–––––––––––––––––––– 2A+B→A2B(overall) Based on this mechanism, determine the rate law for the overall reaction. Express your answer in standard MasteringChemistry format. For example, if the rate law is k[A]3[B]2 type k*[A]^3*[B]^2. View Available Hint(s)

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:30
How many grams of magnesium metal will react completely with 8.3 liters of 5.5m hcl? show all work
Answers: 1


Chemistry, 22.06.2019 18:30
When the chemicals iron sulfide (fes) and hydrochloric acid (hcl) are combined, bubbles appear from the mixture. 1. does the appearance of bubbles indicate a physical or chemical change? 2. why do the bubbles indicate this change? 3. what property is this?
Answers: 1
You know the right answer?
Consider the following multistep reaction: A+B→AB(slow) A+AB→A2B(fast)–––––––––––––––––––– 2A+B→A2B(...
Questions

Advanced Placement (AP), 23.01.2020 22:31





History, 23.01.2020 22:31













Computers and Technology, 23.01.2020 22:31

Health, 23.01.2020 22:31

![Rate=k[A][B]](/tpl/images/0528/6935/27e48.png)
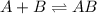 (slow)
(slow)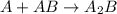 (fast)
(fast)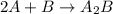 (overall)
(overall)

