Chemistry, 02.03.2020 17:55 shawnr6989
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp. heat of H2O(s) = 2.09 J/g•°C, Sp. heat of H2O(l) = 4.18 J/g•°C, Sp heat of H2O(g) = 2.03 J/g•°C; heat of fus. of H2O(s) = 333 J/g, heat of vap. of H2O(l) = 2260 J/g).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Write two balanced equations 1. dissolving of solid sodium hydroxide in water 2. the reaction of sodium hydroxide solution with hydrochloric acid
Answers: 1

Chemistry, 22.06.2019 14:10
13. a covalent bond between two atoms is likely to be polar if: a. one of the atoms is much more electronegative than the other. b. the two atoms are equally electronegative. c. the two atoms are of the same element. d. the bond is part of a tetrahedrally shaped molecule. e. one atom is an anion.
Answers: 1

Chemistry, 23.06.2019 00:40
To prevent the presence of air, noble gases are placed over highly reactive chemicals to act as inert "blanketing" gases. a chemical engineer places a mixture of noble gases consisting of 4.37 g of he, 13.36 g of ne, and 36.65 g of kr in a piston-cylinder assembly at stp. calculate the partial pressure in torr of kr.
Answers: 1

You know the right answer?
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp...
Questions

History, 04.10.2020 01:01

Mathematics, 04.10.2020 01:01

English, 04.10.2020 01:01



Biology, 04.10.2020 01:01







Mathematics, 04.10.2020 01:01



English, 04.10.2020 01:01

Social Studies, 04.10.2020 01:01

Mathematics, 04.10.2020 01:01

Biology, 04.10.2020 01:01

History, 04.10.2020 01:01

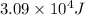
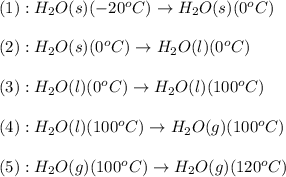
![\Delta H=[m\times c_{p,s}\times (T_{final}-T_{initial})]+m\times \Delta H_{fusion}+[m\times c_{p,l}\times (T_{final}-T_{initial})]+m\times \Delta H_{vap}+[m\times c_{p,g}\times (T_{final}-T_{initial})]](/tpl/images/0530/2584/4a4bb.png)
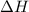 = heat required for the reaction
= heat required for the reaction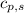 = specific heat of solid water or ice =
= specific heat of solid water or ice = 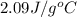
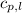 = specific heat of liquid water =
= specific heat of liquid water = 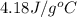
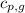 = specific heat of gaseous water =
= specific heat of gaseous water = 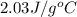
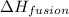 = enthalpy change for fusion =
= enthalpy change for fusion = 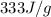
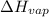 = enthalpy change for vaporization =
= enthalpy change for vaporization = 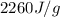
![\Delta H=[10.0g\times 2.09J/g^oC\times (0-(-20))^oC]+10.0g\times 333J/g+[10.0g\times 4.18J/g^oC\times (100-0)^oC]+10.0g\times 2260J/g+[10.0g\times 2.03J/g^oC\times (120-100)^oC]](/tpl/images/0530/2584/295b1.png)