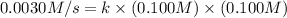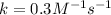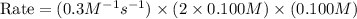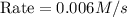Chemistry, 03.03.2020 02:24 batmanmarie2004
When the concentrations of CH 3 Br and NaOH are both 0.100 M, the rate of the reaction is 0.0030 M/s. What is the rate of the reaction if the concentration of CH 3 Br is doubled?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 16:00
1. an experiment in your science class lists the materials needed for the lab. it is your job, as a lab partner, to measure out 25 ml of distilled water and 2.5 grams of magnesium. what lab measuring tools would you choose to measure each substance and how would you use each tool to get the correct amounts? be sure to describe the process you would follow step-by-step. (5 points) 2.which of the following is an si base unit for measuring mass? (2 points) ampere gram meter pound 3.which of the following is an si base unit for time? (2 points) decades hours minutes seconds 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer 4.which of the following tools should a scientist use to measure an object in milligrams? (2 points) graduated cylinder pan balance tape measure thermometer. 5.a pencil beside a metric ruler. the ruler is scaled from 1 centimeter to 10 centimeters, with markings for millimeters between each number. one end of the pencil is beside the 0 on the ruler, and the pencil point is beside the 5. which of the following measurements is accurate but not precise? (2 points) 5 mm 5 cm 50 mm 50 cm 6. which of the following prefixes represents the largest value? (2 points) giga hector kilo milli 7. which of the following types of graphs is best for plotting the percentages of a whole value in a data set? (2 points) bar graph circle graph histogram line graph
Answers: 1

Chemistry, 23.06.2019 01:30
At a certain temperature the rate of this reaction is first order in hi with a rate constant of : 0.0632s2hig=h2g+i2g suppose a vessel contains hi at a concentration of 1.28m . calculate how long it takes for the concentration of hi to decrease to 17.0% of its initial value. you may assume no other reaction is important. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 08:00
If the solubility of a gas in water is 1.22 g/l at 2.75 atm, what is its solubility (in g/l) at 1.0 atm?
Answers: 1
You know the right answer?
When the concentrations of CH 3 Br and NaOH are both 0.100 M, the rate of the reaction is 0.0030 M/s...
Questions

Mathematics, 23.04.2020 23:18

Biology, 23.04.2020 23:18

Mathematics, 23.04.2020 23:18





Mathematics, 23.04.2020 23:18

Mathematics, 23.04.2020 23:18




History, 23.04.2020 23:19

Mathematics, 23.04.2020 23:19

Mathematics, 23.04.2020 23:19

Mathematics, 23.04.2020 23:19




 is doubled is, 0.006 M/s
is doubled is, 0.006 M/s
 are the reactants.
are the reactants.![\text{Rate}=k[CH_3Br][NaOH]](/tpl/images/0531/5144/64424.png)
![[CH_3Br]](/tpl/images/0531/5144/0572a.png) = concentration of
= concentration of ![[NaOH]](/tpl/images/0531/5144/119b0.png) = concentration of
= concentration of