
Chemistry, 17.03.2020 00:57 gracie6313
You take an aspirin tablet (a compound consisting solely of carbon, hydrogen, and oxygen) with a mass of 1.00 g, burn it in air, and collect 2.20 g of carbon dioxide and 0.400 g water. The molar mass of aspirin is between 170 and 190 g/mol. The molecular formula of aspirin is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Adrop of acetone (nail polish remover) has a mass of 35 mg and a density of 0.788 g/cm3. what is its volume in cubic centimeters?
Answers: 3

Chemistry, 22.06.2019 14:00
What is the ph of a solution that has a hydrogen ion concentration of 1.0 * 10 -9 m?
Answers: 2

Chemistry, 22.06.2019 21:30
Achemical reaction is done in the setup shown, resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 1

Chemistry, 23.06.2019 06:10
2. what two items do autotrophs take from the environment to produce their food? 3. what are the two items that are released during transpiration from leaves? 4. what are the two membranes of the system? a.what are the two stages of photosynthesis? what are the two parts of photosynthesis?
Answers: 2
You know the right answer?
You take an aspirin tablet (a compound consisting solely of carbon, hydrogen, and oxygen) with a mas...
Questions



Mathematics, 14.02.2020 19:56


History, 14.02.2020 19:57

Computers and Technology, 14.02.2020 19:57











Mathematics, 14.02.2020 19:57

Mathematics, 14.02.2020 19:57


Health, 14.02.2020 19:57

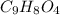
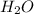 = 0.400 g /18 g/mol = 0.0222 moles
= 0.400 g /18 g/mol = 0.0222 moles
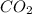 = 2.20 g /44.01 g/mol = 0.05 moles
= 2.20 g /44.01 g/mol = 0.05 moles


