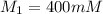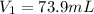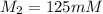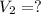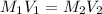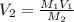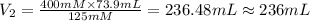A chemist must dilute of aqueous sodium carbonate solution until the concentration falls to . He'll do this by adding distilled water to the solution until it reaches a certain final volume. Calculate this final volume, in milliliters. Be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 01:20
Match the acid base pairs by arranging the acid name with the conjugate base formula. hydrogen carbonate hydrogen phosphate carbonic acid read water sulfuric acid phosphoric acid a. co32- b. hso4- c. hco3- d. po43- e. h2po4- f. oh-
Answers: 1

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2
You know the right answer?
A chemist must dilute of aqueous sodium carbonate solution until the concentration falls to . He'll...
Questions



Advanced Placement (AP), 01.11.2020 20:30



Business, 01.11.2020 20:30

Mathematics, 01.11.2020 20:30

Arts, 01.11.2020 20:30


Mathematics, 01.11.2020 20:30



Mathematics, 01.11.2020 20:40

English, 01.11.2020 20:40

English, 01.11.2020 20:40




Mathematics, 01.11.2020 20:40

Mathematics, 01.11.2020 20:40