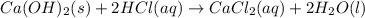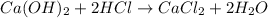Chemistry, 19.03.2020 18:30 idontknow1993
A sample of 8.11 g 8.11 g of solid calcium hydroxide is added to 38.0 mL 38.0 mL of 0.410 M 0.410 M aqueous hydrochloric acid. Write the balanced chemical equation for the reaction. Physical states are optional. chemical equation:

Answers: 1


Another question on Chemistry


Chemistry, 21.06.2019 20:40
If equal masses of the listed metals were collected , which would have a greatest volume ? a. aluminum 2.70,b.zinc7.14,c.copper 8.92,d.lead 11.34
Answers: 2

Chemistry, 22.06.2019 09:00
Particles vibrate in a rigid structure and do not move relative to their neighbors.
Answers: 1

Chemistry, 22.06.2019 09:30
Which ocean zone has the most abundant primary producer and why a) the abyssopelagic zone ,du to the absence of light and cold water temperatureb) the bathypelagic zone, due to the absence of light and cold water temperaturec) the mesopelagic zone ,due to uts high light availability and warm water temperature d) the epipelagic zone,due to its high light availability and warm water temperature
Answers: 3
You know the right answer?
A sample of 8.11 g 8.11 g of solid calcium hydroxide is added to 38.0 mL 38.0 mL of 0.410 M 0.410 M...
Questions


Mathematics, 22.08.2019 03:30


Mathematics, 22.08.2019 03:30

English, 22.08.2019 03:30




History, 22.08.2019 03:30


Mathematics, 22.08.2019 03:30

Health, 22.08.2019 03:30




Chemistry, 22.08.2019 03:30



History, 22.08.2019 03:30

Mathematics, 22.08.2019 03:30