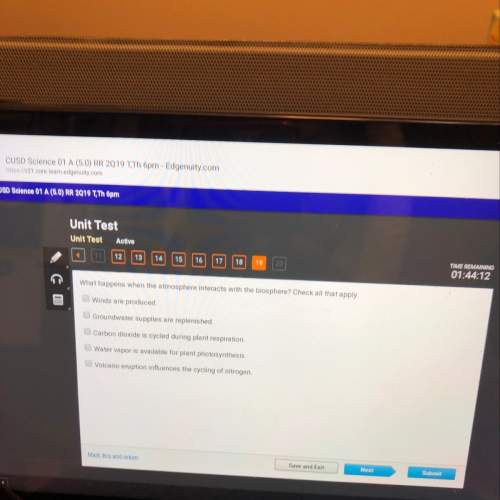
Chemistry, 19.03.2020 23:31 ericgalo808
A chemist fills a reaction vessel with 0.654 g silver chromate (Ag, CrO4) solid, 0.840 M silver aqueous solution, and 0.688 M chromate aqueous solution at a temperature of 25.0° C. Under these conditions, calculate the reaction free energy AG for the following chemical reaction:
Ag2CrO4(s) ---> 2Ag+ (aq) + (CrO4)2- (aq).

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:00
The agent of mechanical weathering in which rock is worn away by the grinding action of other rock particles is call
Answers: 1


Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1
You know the right answer?
A chemist fills a reaction vessel with 0.654 g silver chromate (Ag, CrO4) solid, 0.840 M silver aque...
Questions