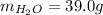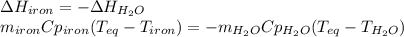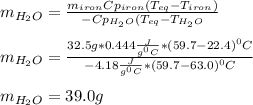Chemistry, 20.03.2020 01:28 ghari112345
A 32.5 g iron rod, initially at 22.4 ∘C, is submerged into an unknown mass of water at 63.0 ∘C, in an insulated container. The final temperature of the mixture upon reaching thermal equilibrium is 59.7 ∘C. Part A What is the mass of the water?

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 15:40
Describe in detail the melting point behavior of the 80: 20 benzoic acid-mandelic acid mixture
Answers: 3

Chemistry, 21.06.2019 21:00
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 21.06.2019 23:00
A100-watt light bulb radiates energy at a rate of 100 j/s. (the watt, a unit of power or energy over time, is defined as 1 j/s.) if all of the light emitted has a wavelength of 525 nm , how many photons are emitted per second?
Answers: 1

You know the right answer?
A 32.5 g iron rod, initially at 22.4 ∘C, is submerged into an unknown mass of water at 63.0 ∘C, in a...
Questions



Mathematics, 09.11.2019 15:31

Mathematics, 09.11.2019 15:31



Mathematics, 09.11.2019 15:31



Mathematics, 09.11.2019 15:31

English, 09.11.2019 15:31

Mathematics, 09.11.2019 15:31

Mathematics, 09.11.2019 15:31

Social Studies, 09.11.2019 15:31

History, 09.11.2019 15:31

Biology, 09.11.2019 15:31

Mathematics, 09.11.2019 15:31