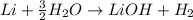Anwser keyClick on Alkali Metals (left bar) and answer the following questions. a. What is the group number? . Are these metals reactive? b. Do these metals occur freely in nature? c. How many electrons are in their outer shell? d. What are the three characteristics of ALL metals? . e. Are these metals soft or hard? f. Name the two most reactive elements in this group? And g. What happens when they are exposed to water? . wab quest answer key

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 08:30
Identify one disadvantage to each of the following models of electron configuration: -dot structures -arrow and line diagrams -written electron configurations type in your answer below. (answer) -dot structures do not show the distribution of electrons in orbitals and take up a lot of space. -arrow and line diagrams take up a lot of space and make it difficult to count electrons. -written configurations make it easy to lose count of electrons and do not show the distribution of electrons in orbitals.
Answers: 3



Chemistry, 22.06.2019 22:30
Consider a culture medium on which only gram-positive organisms such as staphylococcus aureus colonies can grow due to an elevated nacl level. a yellow halo surrounds the growth, indicating the bacterium fermented a sugar in the medium, decreasing the ph as a result and changing the color of a ph indicator chemical. this type of medium would be referred to as a differential and enrichment culture.
Answers: 2
You know the right answer?
Anwser keyClick on Alkali Metals (left bar) and answer the following questions. a. What is the group...
Questions

Advanced Placement (AP), 08.02.2021 18:40

English, 08.02.2021 18:40













Mathematics, 08.02.2021 18:40

Mathematics, 08.02.2021 18:40

Mathematics, 08.02.2021 18:40

English, 08.02.2021 18:40