
Chemistry, 30.03.2020 21:09 rvkanneh1713
(4) After addition of hydrochloric acid, a student removes the lower aqueous layer, extracts the aqueous phase with diethyl ether (0.5 mL), dries the diethyl ether layer with sodium sulfate and pours the ~0.5 mL of diethyl ether into an Erlenmeyer flask, rinsing the drying agent with more diethyl ether and pouring into the flask. Upon evaporation of the diethyl ether, the student is surprised to see no crystals present to recrystallize. Assuming Parts 1–3 were completed successfully, explain where the student lost their product.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 16:00
As changes in energy levels of electrons increase, the frequencies of atomic line spectra they emit
Answers: 2

Chemistry, 22.06.2019 16:50
What is conserved in the reaction shown below? h2(g) + cl2 (g) --> 2hcl(g)a. mass onlyb. mass and moles onlyc. mass, moles, and molecules onlyd. mass, moles, molecules, and volume
Answers: 2
You know the right answer?
(4) After addition of hydrochloric acid, a student removes the lower aqueous layer, extracts the aqu...
Questions

Mathematics, 05.05.2020 16:14





Physics, 05.05.2020 16:14

Mathematics, 05.05.2020 16:14


Mathematics, 05.05.2020 16:14

Medicine, 05.05.2020 16:14


History, 05.05.2020 16:14








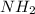 in the compound which have a tendency of forming
in the compound which have a tendency of forming 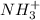 salt.
salt.

