Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:30
Which formula can be used to calculate the molar mass of hydrogen peroxide
Answers: 1

Chemistry, 22.06.2019 13:00
Jose and eric were given four samples in lab. the results of their analysis are shown in the table. based on the data they collected, which sample is most likely a metal?
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1
You know the right answer?
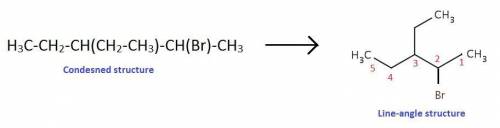
Draw the line-angle structure for the given condensed structure, and map the carbon atoms in the con...
Questions


Biology, 13.03.2020 00:13


Mathematics, 13.03.2020 00:14



Mathematics, 13.03.2020 00:14



Mathematics, 13.03.2020 00:14



Geography, 13.03.2020 00:14


Mathematics, 13.03.2020 00:15

Computers and Technology, 13.03.2020 00:15


Mathematics, 13.03.2020 00:15


History, 13.03.2020 00:15