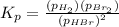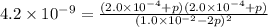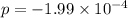Chemistry, 04.04.2020 20:04 nell1234565
At an elevated temperature, Kp=4.2 x 10^-9 for the reaction 2HBr (g)---> +H2(g) + Br2 (g). If the initial partial pressures of HBr, H2, and Br2 are 1.0 x 10^-2 atm, 2.0 x 10^-4 atm, and 2.0 x 10^-4 atm, respecivtely, what is the equilbrium partial pressure of H2?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:00
Which of the following would not diffuse through the plasma membrane by means of simple diffusion? 1 oxygen 2 glucose 3 a steroid hormone 4 a lipid soluble vitamin
Answers: 3

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 20:00
State one important difference between a physical change and a chemical change?
Answers: 1
You know the right answer?
At an elevated temperature, Kp=4.2 x 10^-9 for the reaction 2HBr (g)---> +H2(g) + Br2 (g). If the...
Questions




Computers and Technology, 01.11.2019 07:31

History, 01.11.2019 07:31

History, 01.11.2019 07:31

Mathematics, 01.11.2019 07:31



Biology, 01.11.2019 07:31

Mathematics, 01.11.2019 07:31


Spanish, 01.11.2019 07:31


Mathematics, 01.11.2019 07:31


History, 01.11.2019 07:31

History, 01.11.2019 07:31


Mathematics, 01.11.2019 07:31

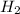 at equilibrium is, 1.0 × 10⁻⁶
at equilibrium is, 1.0 × 10⁻⁶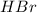 =
= 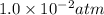
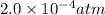
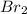 =
= 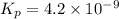
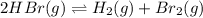
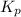 for the reaction will be:
for the reaction will be: