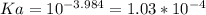Chemistry, 07.04.2020 23:52 brendaesme
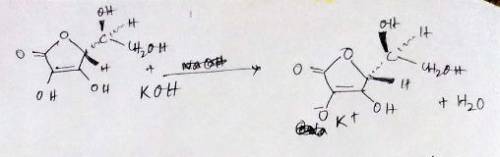
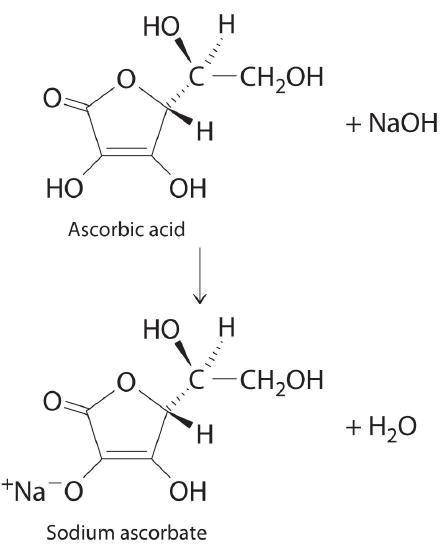
A 0.552-g sample of ascorbic acid was dissolved in water to a total volume of 0.20 mL and titrated with 0.1103 M KOH. The equivalence point occurred at 28.42 mL. The pH of the solution at 10.0mL of added base was 3.72. From this data, determine the molar rmass and Ka for vitamin C.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 22:30
What relationship exists between an enzyme and a catalyst?
Answers: 1
You know the right answer?
A 0.552-g sample of ascorbic acid was dissolved in water to a total volume of 0.20 mL and titrated w...
Questions

Biology, 10.02.2021 23:00





English, 10.02.2021 23:00


Mathematics, 10.02.2021 23:00

Mathematics, 10.02.2021 23:00

Chemistry, 10.02.2021 23:00







English, 10.02.2021 23:00



English, 10.02.2021 23:00