
Chemistry, 08.04.2020 01:53 HecptyAura
Which statement is correct? Group of answer choices none of these H2O is linear. The bonds in LiF have a more covalent character than those in F2. The diatomic molecule Cl2 is an example of a polar molecule. The molecule ClO2 cannot be accurately described by a Lewis structure consistent with the octet rule.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2

Chemistry, 23.06.2019 04:00
What is the volume of 2.5 moles of nitrogen gas (n2)at standard temperature and pressure (stp)?
Answers: 1

Chemistry, 23.06.2019 07:00
Under what conditions will a gas be most likely to exhibit the ideal gas properties predicted by the ideal gas law? 1)high pressures and high temperature, because particles are forced closer together with higher kinetic energy, so intermolecular forces of attraction are weaker 2)high pressure and low temperature, because particles are forced closer together and moving slower, so the volume of the particles is less significant 3) low pressure and high temperature, because particles are spread farther apart and moving faster, so the intermolecular forces of attraction are weaker 4)low pressure and low temperature, because particles are spread farther apart with lower kinetic energy, so the volume of the particles is less significant
Answers: 2
You know the right answer?
Which statement is correct? Group of answer choices none of these H2O is linear. The bonds in LiF ha...
Questions


Mathematics, 02.09.2019 06:10

History, 02.09.2019 06:10


History, 02.09.2019 06:10

Mathematics, 02.09.2019 06:10

Mathematics, 02.09.2019 06:10


Mathematics, 02.09.2019 06:10


English, 02.09.2019 06:10

Biology, 02.09.2019 06:10



World Languages, 02.09.2019 06:10


Geography, 02.09.2019 06:10



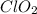 cannot be accurately described by a Lewis structure consistent with the octet rule, is true.
cannot be accurately described by a Lewis structure consistent with the octet rule, is true.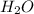 and since it contains electronegative oxygen atom which also has 2 lone pair of electrons. Hence, lone pair-bond pair repulsion occurs due to which water has a bent shape structure.
and since it contains electronegative oxygen atom which also has 2 lone pair of electrons. Hence, lone pair-bond pair repulsion occurs due to which water has a bent shape structure.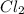 is a non-polar compound.
is a non-polar compound.

