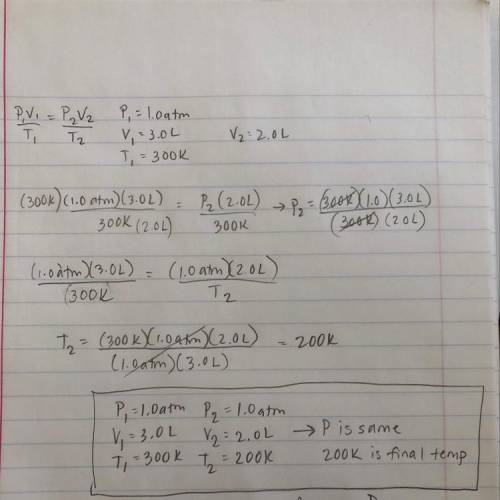Chemistry, 08.04.2020 21:09 blueval3tine
The volume of a gas is 3.0 L, the pressure is 1.0 atm, and the temperature is 300 K. A chemist changes one factor while keeping another constant so that the new volume is 2.0 L. Which of the following could be the new conditions? (5 points)

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 04:50
The name of the ion, s2-, is: sulfurous ion sulfide ion sulfur ion sulfate ion
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 20:10
What would happen to a volleyball left outside in the winter? o o o o a. it would expand. b. it would lose air. c. it would shrink. d. it would explode.
Answers: 2
You know the right answer?
The volume of a gas is 3.0 L, the pressure is 1.0 atm, and the temperature is 300 K. A chemist chang...
Questions

Social Studies, 15.11.2019 20:31

Computers and Technology, 15.11.2019 20:31


Social Studies, 15.11.2019 20:31



Mathematics, 15.11.2019 20:31






Mathematics, 15.11.2019 20:31

World Languages, 15.11.2019 20:31

Mathematics, 15.11.2019 20:31

Social Studies, 15.11.2019 20:31