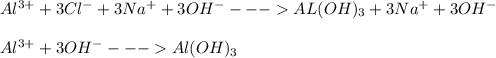Chemistry, 19.04.2020 06:26 TGJuuzouSuzuya
Write the balanced NET ionic equation for the reaction when AlCl₃ and NaOH are mixed in aqueous solution. If no reaction occurs, write only NR.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2

Chemistry, 23.06.2019 04:20
The graph shows one consequence of urban sprawl. how did urban sprawl contribute to the change in biodiversity
Answers: 2

Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
Write the balanced NET ionic equation for the reaction when AlCl₃ and NaOH are mixed in aqueous solu...
Questions

History, 23.09.2020 01:01




Biology, 23.09.2020 01:01



Social Studies, 23.09.2020 01:01


Mathematics, 23.09.2020 01:01

Mathematics, 23.09.2020 01:01


Mathematics, 23.09.2020 01:01


Biology, 23.09.2020 01:01


Social Studies, 23.09.2020 01:01

Chemistry, 23.09.2020 01:01

Advanced Placement (AP), 23.09.2020 01:01

Mathematics, 23.09.2020 01:01