
A chemist designs a galvanic cell that uses these two half-reactions:
O2 (g) + 4H+(aq) + 4e− → 2H2O (l) Eo =+1.23V
Zn+2 (aq) + 2e− → Zn(s) Eo=−0.763V
Answer the following questions about this cell.
Write a balanced equation for the half-reaction that happens at the cathode.
Write a balanced equation for the half-reaction that happens at the anode.
Write a balanced equation for the overall reaction that powers the cell. Be sure the reaction is spontaneous as written. Do you have enough information to calculate the cell voltage under standard conditions

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
If you add 10ml of hot water to 10ml of cold water and the change in tempature 8°c calculate how much energy is gained by the cold water
Answers: 1

Chemistry, 22.06.2019 13:00
What is the mass of 2.00 l of an intravenous glucose solution with a density of 1.15 g/ml?
Answers: 2


Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
A chemist designs a galvanic cell that uses these two half-reactions:
O2 (g) + 4H+(aq) +...
O2 (g) + 4H+(aq) +...
Questions








English, 27.02.2020 02:02





Biology, 27.02.2020 02:03

Mathematics, 27.02.2020 02:03



Mathematics, 27.02.2020 02:03


Mathematics, 27.02.2020 02:03

Mathematics, 27.02.2020 02:03

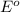 reduction potential will always get reduced and will undergo reduction reaction.
reduction potential will always get reduced and will undergo reduction reaction. 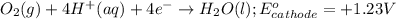
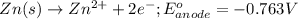 ( × 2)
( × 2)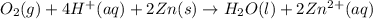
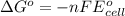
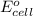 of the reaction, we use the equation:
of the reaction, we use the equation: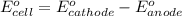
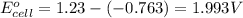
![E_{cell}=E^o_{cell}-\frac{0.059}{n}\log \frac{[Zn^{2+}]^2}{[H^{+}]^4\times p_{O_2}}](/tpl/images/0645/2279/2e91a.png)


