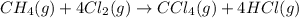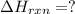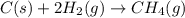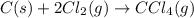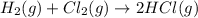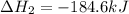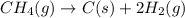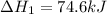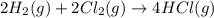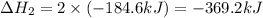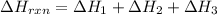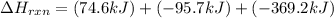Chemistry, 12.06.2020 04:57 yurionice42069
PLZ SEE ATTACHED AND I WOULD REALLY APPRECIATE IT! ANYONE GOOD WITH CHEM
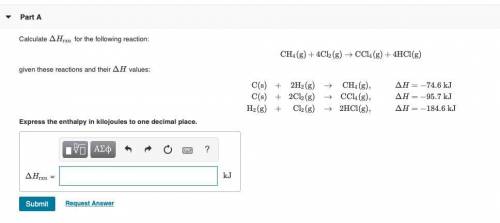

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 00:20
4. propanol and isopropanol are isomers. this means that they have a) the same molecular formula but different chemical properties. b) different molecular formulas but the same chemical properties. c) the same molecular formula and the same chemical properties. d) the same molecular formula but represent different states of the compound
Answers: 3

You know the right answer?
PLZ SEE ATTACHED AND I WOULD REALLY APPRECIATE IT! ANYONE GOOD WITH CHEM
...
...
Questions


History, 26.08.2019 20:30


Computers and Technology, 26.08.2019 20:30

Geography, 26.08.2019 20:30


Chemistry, 26.08.2019 20:30

Social Studies, 26.08.2019 20:30

Biology, 26.08.2019 20:30



Social Studies, 26.08.2019 20:30

English, 26.08.2019 20:30

Mathematics, 26.08.2019 20:30

History, 26.08.2019 20:30

Biology, 26.08.2019 20:30

English, 26.08.2019 20:30

Geography, 26.08.2019 20:30

Biology, 26.08.2019 20:30

Mathematics, 26.08.2019 20:30

 for the reaction is, -390.3 kJ
for the reaction is, -390.3 kJ