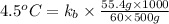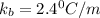Chemistry, 21.06.2020 06:57 ethanmatthews2336
A certain substance X condenses at a temperature of 120.7 degree C. But if a 500, g sample of X is prepared with 55.4 g of urea (NH_2)_2 CO) dissolved in it, the sample is found to have a condensation point of 125.2 degree C instead. Calculate the molal boiling point elevation constant K_b of X. Round your answer to 2 significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:00
Which of the following are good traits of a hypothesis? it will be able to be testedit can predict an outcomeit will explain the observationsall of these
Answers: 2

Chemistry, 22.06.2019 18:30
When a device is used in a circuit in which the voltage is 81 v the current flowing through the device is 3 a what is the resistance of the device
Answers: 2


Chemistry, 23.06.2019 01:00
Heat energy, carbon dioxide, and water are released through which process? a. photosynthesis b. depolymerization c. digestion d. cellular respiration
Answers: 1
You know the right answer?
A certain substance X condenses at a temperature of 120.7 degree C. But if a 500, g sample of X is p...
Questions


History, 10.01.2021 06:20


Mathematics, 10.01.2021 06:20

Mathematics, 10.01.2021 06:20

Mathematics, 10.01.2021 06:20

Computers and Technology, 10.01.2021 06:20

Arts, 10.01.2021 06:20

Advanced Placement (AP), 10.01.2021 06:20



Mathematics, 10.01.2021 06:20


Mathematics, 10.01.2021 06:20






Mathematics, 10.01.2021 06:30

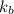 of X is
of X is 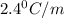
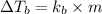
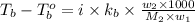
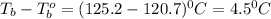
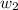 = mass of solute (urea) = 55.4 g
= mass of solute (urea) = 55.4 g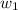 = mass of solvent X = 500 g
= mass of solvent X = 500 g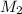 = molar mass of solute (urea) = 60 g/mol
= molar mass of solute (urea) = 60 g/mol