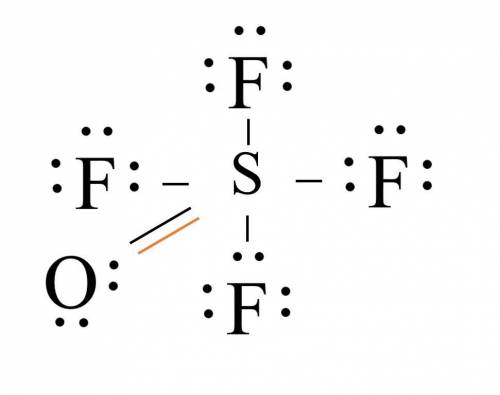Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 22.06.2019 20:30
We are hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 2

Chemistry, 23.06.2019 00:30
Five different substances are given to you to be dissolved in water. which substances are most likely to undergo dissolution in water? check all that apply. view available hint(s) check all that apply. sodium fluoride, naf octane, c8h18 propanol, ch3ch2ch2oh potassium iodide, ki benzene, c6h6
Answers: 1
You know the right answer?
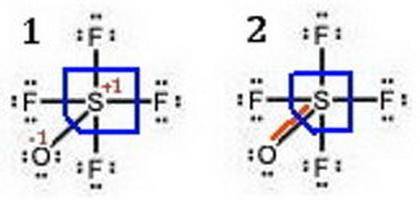
Given that S is the central atom, draw a Lewis structure of OSF4 in which the formal charges of all...
Questions



Computers and Technology, 29.06.2019 04:00




Social Studies, 29.06.2019 04:00


Social Studies, 29.06.2019 04:00






Social Studies, 29.06.2019 04:00





Social Studies, 29.06.2019 04:00

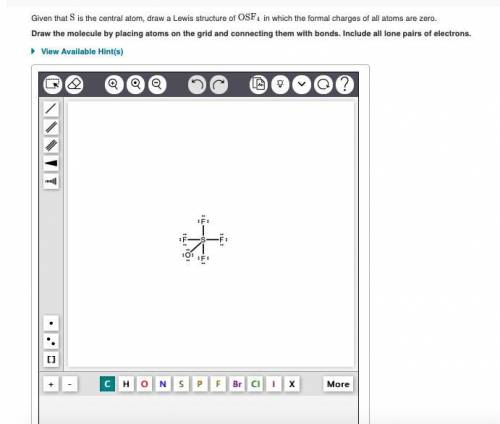
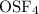 , the formal charge has been assigned zero with the bond formation resulting in the valence electrons and bonding electrons being equal.
, the formal charge has been assigned zero with the bond formation resulting in the valence electrons and bonding electrons being equal.