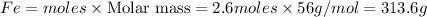The following balanced equation describes the reduction of iron(III) oxide to molten iron within a blast furnace: Fe2O3(s) + 3CO(g) ---> 2Fe(l) + 3CO2(g) Steve inserts 450. g of iron(III) oxide and 260. g of carbon monoxide into the blast furnace. After cooling the pure liquid iron, Steve determines that he has produced 288g of iron ingots. What is the theoretical yield of liquid iron, in grams? Just enter a numerical value. Do not enter units.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 05:50
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3 -> a2cr(no3)3 + a3ag2so4
Answers: 1

Chemistry, 22.06.2019 12:30
Sodium sulfate dissolves as follows: na2so4(s) → 2na+(aq) + so42- (aq). how many moles of na2so4 are required to make 1.0 l of solution in which the na concentration is 0.10 m?
Answers: 2

Chemistry, 22.06.2019 14:30
Connect the whole numbers on the periodic table to indicate what they represent?
Answers: 3
You know the right answer?
The following balanced equation describes the reduction of iron(III) oxide to molten iron within a b...
Questions


Biology, 22.07.2019 19:30



Social Studies, 22.07.2019 19:30





History, 22.07.2019 19:30






Computers and Technology, 22.07.2019 19:30

History, 22.07.2019 19:30

Social Studies, 22.07.2019 19:30


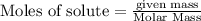
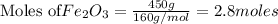
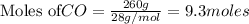
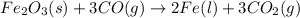
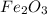 require 3 moles of
require 3 moles of 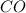
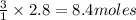 of
of 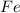
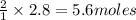 of
of