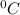Chemistry, 04.08.2020 14:01 aislynrae22
A 1.0 kg object absorbs 1,303 J of heat energy and experiences a temperature increase of 5.2∘C. What is the object’s specific heat, in joules per gram-degree celsius? Report your answer with the correct number of significant figures.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:50
Consider the equilibrium system: 2icl(s) ⇄ i2(s) + cl2(g) which of the following changes will increase the total amount of of cl2 that can be produced? all of the listed answers are correct decreasing the volume of the container removing the cl2 as it is formed adding more icl(s) removing some of the i2(s)
Answers: 1

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 22.06.2019 17:30
Oil rich countries in the middle east cover about 4% of earths total land area but prossess about 48% of the worlds known oil reserves what is the main reason for high concentration of reserves in this part of the world
Answers: 3

Chemistry, 22.06.2019 21:30
What is happening when the water inside a kettle heats up and begins to boil
Answers: 1
You know the right answer?
A 1.0 kg object absorbs 1,303 J of heat energy and experiences a temperature increase of 5.2∘C. What...
Questions

Mathematics, 09.02.2021 05:00



Physics, 09.02.2021 05:00










Mathematics, 09.02.2021 05:00

History, 09.02.2021 05:00



Mathematics, 09.02.2021 05:00