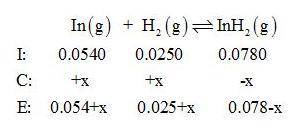Chemistry, 05.08.2020 16:01 XxDonaldTrumpxX452
Gaseous indium dihydride is formed from the elements at elevated temperature:
In(g)+H2(g)⇌InH2(g),Kp=1.48 at 973 K
The partial pressures measured in a reaction vessel are
PIn =0.0540atm
PH2= 0.0250atm
PInH2 =0.0780atm
Calculate Qp and give equal partial pressure for In, H2, and InH2.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 11:00
Imagine that twenty i.u.’s of enzyme z were catalyzing the above reaction for one minute, under vmaxconditions, in a 3.00 ml assay volume. the assay is buffered with 20 mm phosphate buffer, ph 7.60. what will the ph be at the end of that one minute?
Answers: 2

Chemistry, 22.06.2019 12:20
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.
Answers: 3

Chemistry, 22.06.2019 16:40
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3
You know the right answer?
Gaseous indium dihydride is formed from the elements at elevated temperature:
In(g)+H2(g)⇌InH2(g),...
Questions


Biology, 24.09.2019 03:30

English, 24.09.2019 03:30






Mathematics, 24.09.2019 03:30


Mathematics, 24.09.2019 03:30

Health, 24.09.2019 03:30

Mathematics, 24.09.2019 03:30



Biology, 24.09.2019 03:30



Mathematics, 24.09.2019 03:30

Arts, 24.09.2019 03:30