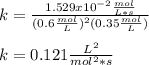Chemistry, 03.09.2020 01:01 milkshakegrande101
Hydrogen reacts with nitrogen monoxide to form dinitrogen monoxide (laughing gas) according to the equation: H2O(g) + 2NO(g) rightarrow O + H2O(g)NO (M) 0.30 0.60 0.60H2 (M) 0.35 0.35 0.70Rate (M/L/s) 3.822 x 103 1.529 x 10-2 3.058 x 10-2 A) Determine the rate law according to the following data. B) Determine the rate constant (in mol-2. L2. s-1).C) Determine the orders with respect to each reactant.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 22.06.2019 18:00
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1


Chemistry, 23.06.2019 00:30
How many moles of co2 are produced during the complete combustion of 3.6 moles of c2h6
Answers: 1
You know the right answer?
Hydrogen reacts with nitrogen monoxide to form dinitrogen monoxide (laughing gas) according to the e...
Questions




English, 06.07.2019 22:00

History, 06.07.2019 22:00

English, 06.07.2019 22:00


Business, 06.07.2019 22:00



Physics, 06.07.2019 22:00

Mathematics, 06.07.2019 22:00



Mathematics, 06.07.2019 22:00

Mathematics, 06.07.2019 22:00

Mathematics, 06.07.2019 22:00

Physics, 06.07.2019 22:00

Spanish, 06.07.2019 22:00

![r=k[NO]^2[H_2]](/tpl/images/0739/2059/50be7.png)
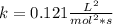
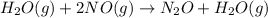
![3.822x10^{-3}=k[0.3]^m[0.35]^n\\\\1.529x10^{-2}=k[0.6]^m[0.35]^n\\\\3.058x10^{-2}=k[0.6]^m[0.7]^n](/tpl/images/0739/2059/1a97a.png)
![\frac{3.822x10^{-3}}{1.529x10^{-2}} =\frac{[0.3]^m[0.35]^n}{[0.6]^m[0.35]^n} \\\\0.25=(0.5)^m\\\\m=\frac{log(0.25)}{log(0.5)} \\\\m=2](/tpl/images/0739/2059/4863a.png)
![\frac{1.529x10^{-2}}{3.058x10^{-2}} =\frac{[0.6]^2[0.35]^n}{[0.6]^2[0.7]^n} \\\\0.5=(0.5)^n\\\\n=\frac{log(0.5)}{log(0.5)}\\ \\n=1](/tpl/images/0739/2059/67e47.png)