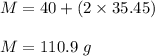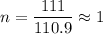Chemistry, 19.09.2020 01:01 SunsetPrincess
A certain compound contains 4.0 g of calcium and 7.1 g of chlorine. Its relative molecular mass is 111. Find its empirical and molecular formulas

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 07:00
Achemist wants to extract copper metal from copper chloride solution. the chemist places 0.50 grams of aluminum foil in a solution containing 0.75 grams of copper (ii) chloride. a single replacement reaction takes place. (ii) chloride. a single replacement reaction takes place. which statement explains the maximum amount of copper that the chemist can extract using this reaction? a) approximately 0.36 grams, because copper (ii) chloride acts as a limiting reactant b) approximately 1.8 grams, because copper (ii) chloride acts as a limiting reactant c) approximately 0.36 grams, because aluminum acts as a limiting reactant d) approximately 1.8 grams, because aluminum acts as a limiting reactant
Answers: 3

Chemistry, 22.06.2019 14:30
Select all that apply. using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 (s) pb+2(aq) + 2cl -(aq). the concentration of the products yield a ksp of 2.1 x 10-2:
Answers: 2

Chemistry, 23.06.2019 06:10
How much would the freezing point of water decrease if 4 mol of nacl were added to 1 kg of water (kf=1.86 degrees c/(mol/kg) for water and i=2 for nacl a- 7.44 degrees c b- 14.88 c 3.72 d 1.86
Answers: 1
You know the right answer?
A certain compound contains 4.0 g of calcium and 7.1 g of chlorine. Its relative molecular mass is 1...
Questions



Mathematics, 27.03.2020 17:56



Biology, 27.03.2020 17:56


English, 27.03.2020 17:56

Mathematics, 27.03.2020 17:56


Mathematics, 27.03.2020 17:56

Computers and Technology, 27.03.2020 17:56


Biology, 27.03.2020 17:56


Social Studies, 27.03.2020 17:56


Computers and Technology, 27.03.2020 17:56


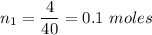 .
.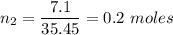 .
.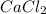 .
.