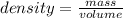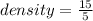Calculate the density of the items below. (D = M/V)
1) M = 15g V = 5.0ml *...

Chemistry, 14.10.2020 02:01 desiree3114
Calculate the density of the items below. (D = M/V)
1) M = 15g V = 5.0ml *

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:50
Which best describes why nh4+ can form an ionic bond with cl-?
Answers: 3

Chemistry, 22.06.2019 03:30
Each pair of clay balls represents to planetesimals if each plane test molluscum pound of the same material and is separated by the same distance which pair experiences the greatest gravitational attraction
Answers: 2

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 07:00
At 450 mm hg a gas has a volume of 760 l, what is its volume at standard pressure
Answers: 2
You know the right answer?
Questions


Biology, 13.10.2019 01:30

Mathematics, 13.10.2019 01:30



Biology, 13.10.2019 01:30




Social Studies, 13.10.2019 01:30

English, 13.10.2019 01:30


Mathematics, 13.10.2019 01:30


Mathematics, 13.10.2019 01:30


Arts, 13.10.2019 01:30

Chemistry, 13.10.2019 01:30

Mathematics, 13.10.2019 01:30