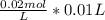Chemistry, 21.10.2020 17:01 cia196785920
Suppose a student needs to standardize a sodium thiosulfate, Na2S2O3,Na2S2O3, solution for a titration experiment. To do so, he or she will react it with a solution of iodine. The student adds a 1.00 mL1.00 mL aliquot of 0.0200 M KIO30.0200 M KIO3 solution to a flask, followed by 3 mL3 mL of distilled water, 0.2 g0.2 g of solid KI, KI, and 1 mL H2SO4.1 mL H2SO4. The student then titrates the solution with sodium thiosulfate solution in order to determine the exact concentration of Na2S2O3.Na2S2O3. The end point of the titration is reached after 0.90 mL0.90 mL of Na2S2O3Na2S2O3 is dispensed from a microburet. What is the concentration of the standard sodium thiosulfate solution?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 08:30
Which metal exist in liquid state and can be cut with knife ?
Answers: 2

Chemistry, 22.06.2019 15:00
Why does a plastic bottle that is sealed at a high altitude change it’s shape when taken to lower altitude
Answers: 2
You know the right answer?
Suppose a student needs to standardize a sodium thiosulfate, Na2S2O3,Na2S2O3, solution for a titrati...
Questions



Health, 25.07.2019 02:00

English, 25.07.2019 02:00


English, 25.07.2019 02:00


English, 25.07.2019 02:00





History, 25.07.2019 02:00

History, 25.07.2019 02:00

Biology, 25.07.2019 02:00

Mathematics, 25.07.2019 02:00


Social Studies, 25.07.2019 02:00

Social Studies, 25.07.2019 02:00