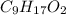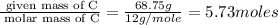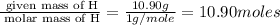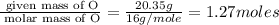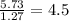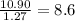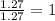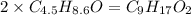Chemistry, 10.10.2019 22:50 tabbydory3366
Determine the number of atoms of each element in the empirical formula of a compound with the following composition:
68.75 percent c, 10.90 percent h, 20.35 percent o.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would be an accurate picture of the earth during the summer time of the northern hemisphere?
Answers: 1

Chemistry, 22.06.2019 17:30
Aroller coaster is traveling at 13 mi./s when you purchase a hill that is 400 m long and down the hill exonerate at 4.0 m/s squared what is the final velocity of the posterior found your answer to the nearest number
Answers: 1

Chemistry, 23.06.2019 02:00
The point along a planet's orbit where it is closest to the sun is called the
Answers: 1
You know the right answer?
Determine the number of atoms of each element in the empirical formula of a compound with the follow...
Questions

History, 15.07.2019 16:00


Mathematics, 15.07.2019 16:00

Mathematics, 15.07.2019 16:00

Business, 15.07.2019 16:00

Mathematics, 15.07.2019 16:00

Biology, 15.07.2019 16:00


Mathematics, 15.07.2019 16:00





Mathematics, 15.07.2019 16:00

Chemistry, 15.07.2019 16:00

English, 15.07.2019 16:00


Mathematics, 15.07.2019 16:00

Mathematics, 15.07.2019 16:00

History, 15.07.2019 16:00