Answers: 3


Another question on Chemistry



Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2
You know the right answer?
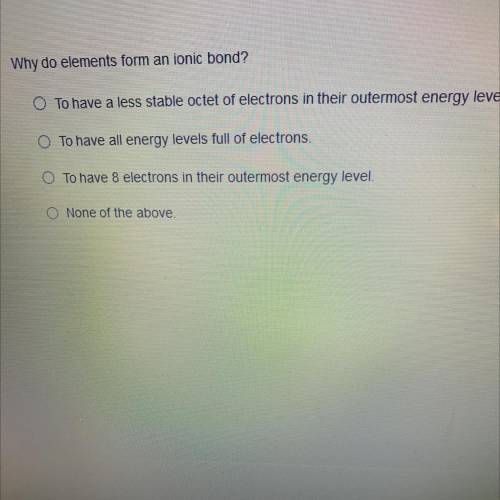
Why do elements form an ionic bond
...
...
Questions



Mathematics, 03.12.2021 20:50

Physics, 03.12.2021 20:50


Mathematics, 03.12.2021 20:50

History, 03.12.2021 20:50

Chemistry, 03.12.2021 20:50

Computers and Technology, 03.12.2021 20:50


Mathematics, 03.12.2021 20:50


Mathematics, 03.12.2021 20:50

Mathematics, 03.12.2021 20:50

Social Studies, 03.12.2021 20:50

Mathematics, 03.12.2021 20:50



Mathematics, 03.12.2021 20:50

Computers and Technology, 03.12.2021 20:50