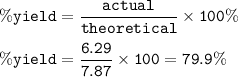At high temperatures, sulfur combines with iron to form brown-black iron(II) sulfide:
Fe(s) + SD-Fes(s)
In an experiment, 5.00 g of iron reacts with excess sulfur to produce 6.29 g of iron(II) sulfide. Calculate the percent yield if the maximum
amount of iron(II) sulfide that can be produced is 7.87 g.
79.796
79.9%
100%
63.5%
125%

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:10
26. of of (aq) by (aq) is . if 50.00 ml of 1.05 m is to 25.00 ml of 1.86 m ,at be? ( no is toina of aof) , h.. (p. ). . .
Answers: 3

Chemistry, 22.06.2019 04:30
What are the three major branches of natural science? • earth and space science, life science, physical science •earth and space science, physical science, chemistry •physical science, life science, chemistry •life science, chemistry, physics
Answers: 1

Chemistry, 22.06.2019 08:30
Which of the following would have less momentum than a 52 kg cheetah running at 10 m/s?
Answers: 2

Chemistry, 22.06.2019 09:00
Chen drew a diagram to compare the ways in which different organisms obtain nitrogen. which label belongs to the area marked z?
Answers: 3
You know the right answer?
At high temperatures, sulfur combines with iron to form brown-black iron(II) sulfide:
Fe(s) + SD-Fe...
Questions

Mathematics, 03.02.2020 07:56


Biology, 03.02.2020 07:56


Chemistry, 03.02.2020 07:56


Mathematics, 03.02.2020 07:56

History, 03.02.2020 07:56

Mathematics, 03.02.2020 07:56







Mathematics, 03.02.2020 07:56

Social Studies, 03.02.2020 07:56

Mathematics, 03.02.2020 07:56


Chemistry, 03.02.2020 07:57