
Chemistry, 03.12.2020 18:40 rjennis002
The following reaction shows sodium carbonate reacting with calcium hydroxide.
Na2CO3 + Ca(OH)2 → 2NaOH + CaCO3
How many grams of NaOH are produced from 20.0 grams of Na2CO3?
(Molar mass of Na = 22.989 g/mol, C = 12.01 g/mol, O = 15.999 g/mol, Ca = 40.078 g/mol, H = 1.008 g/mol) (5 points)
a. 12.2 grams
b. 15.1 grams
c. 24.4 grams
d. 30.2 grams

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:40
Calculate the number of kilojoules to warm 125 g of iron from 23.5°c to 78.0°c.
Answers: 3

Chemistry, 22.06.2019 12:00
Ageochemist examines a piece of metal that he found in the soil. he performs tests to identify the metal from its density, electrical conductivity, and melting point. which statement best describes his investigation? a. he is determining physical properties that are sufficient to identify the metal.b. he is determining chemical properties that are sufficient to identify the metal.c. he is determining physical properties that are insufficient to identify the metal.d. he is determining chemical properties that are insufficient to identify the metal.
Answers: 3

Chemistry, 22.06.2019 12:10
Building glycogen from glucose molecules is an example of
Answers: 3

Chemistry, 22.06.2019 12:30
Which of the following describes a compound? (hint: carbon and oxygen bo a. a piece of pure carbon, containing only carbon atoms b. oxygen gas surrounding a solid piece of carbon c. a substance made of two oxygen atoms for each carbon atom carbon and oxygen atoms mixed without being bonded together
Answers: 1
You know the right answer?
The following reaction shows sodium carbonate reacting with calcium hydroxide.
Na2CO3 + Ca(OH)2 → 2...
Questions

Mathematics, 04.08.2019 21:30





Health, 04.08.2019 21:30

Biology, 04.08.2019 21:30


Social Studies, 04.08.2019 21:30

Arts, 04.08.2019 21:30


Social Studies, 04.08.2019 21:30





Mathematics, 04.08.2019 21:30



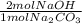 = 0.378 moles NaOH
= 0.378 moles NaOH

