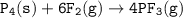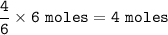Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s)...

Chemistry, 09.12.2020 17:00 tonydeanfbg8706
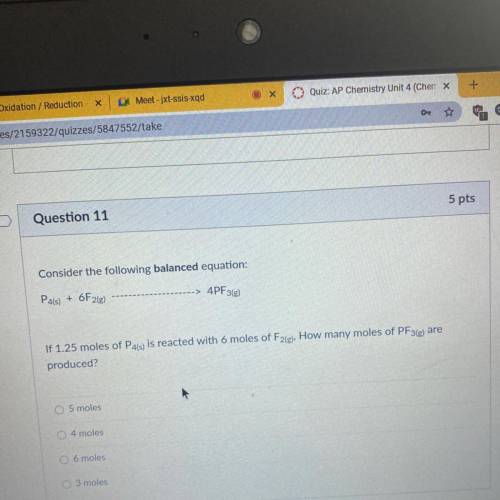
Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s) is reacted with 6 moles of F2(g), How many moles of PF3(e) are
produced?
O 5 moles
4 moles
O 6 moles
O 3 moles

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:30
Which type of stress results when two plates push against one another? a. compression b. tension c. force d. shear
Answers: 1


Chemistry, 22.06.2019 16:10
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 16:40
Let the ed50 of a recreational drug be defined as the amount required for 50% of a test group to feel high or get a buzz. if the ed50 value of ethanol is 470 mg/kg body mass, what dose would a 70 kg party goer need to quickly consume in order to have a 50% chance of getting a buzz? 235 mg 470 mg 32,900 mg 35,000,000 mg
Answers: 3
You know the right answer?
Questions

Advanced Placement (AP), 27.01.2021 14:00




Mathematics, 27.01.2021 14:00

Chemistry, 27.01.2021 14:00

Biology, 27.01.2021 14:00

Arts, 27.01.2021 14:00

Mathematics, 27.01.2021 14:00

Mathematics, 27.01.2021 14:00



Mathematics, 27.01.2021 14:00

History, 27.01.2021 14:00


Mathematics, 27.01.2021 14:00


Mathematics, 27.01.2021 14:00

Mathematics, 27.01.2021 14:00

Computers and Technology, 27.01.2021 14:00