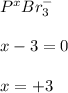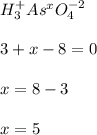Chemistry, 08.01.2021 03:40 kierraware04
g Determine the oxidation state for each of the elements below. ... The oxidation state of ... phosphorus ... in ... phosphorous tribromide PBr3 ... is ... . The oxidation state of nitrogen in nitrogen gas N2 is . The oxidation state of arsenic in arsenic acid H3AsO4 is .

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 07:30
Label a-f based on the table using c for concentrated and d for dilute
Answers: 2

Chemistry, 22.06.2019 11:30
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 23.06.2019 00:30
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3
You know the right answer?
g Determine the oxidation state for each of the elements below. ... The oxidation state of ... phosp...
Questions


Mathematics, 26.04.2021 14:50


Mathematics, 26.04.2021 14:50


History, 26.04.2021 14:50




Mathematics, 26.04.2021 14:50



English, 26.04.2021 14:50



Mathematics, 26.04.2021 14:50

Mathematics, 26.04.2021 14:50