Answers: 1


Another question on Chemistry




Chemistry, 22.06.2019 22:10
Determine the ph of 0.10 m nh3 solution. nh3 is a weak base with a kb equal to 1.8 x 10-5 round answer to nearest whole number.
Answers: 1
You know the right answer?
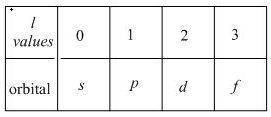
What atomic orbitals are related to a p sublevel?...
Questions

Chemistry, 19.12.2020 23:30

Mathematics, 19.12.2020 23:30


Mathematics, 19.12.2020 23:30

Social Studies, 19.12.2020 23:30


Mathematics, 19.12.2020 23:30





Mathematics, 19.12.2020 23:30

Mathematics, 19.12.2020 23:30


Mathematics, 19.12.2020 23:30

Mathematics, 19.12.2020 23:30

Health, 19.12.2020 23:30

History, 19.12.2020 23:30


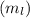 : This quantum number represents the orientation of the orbital in space. The value of
: This quantum number represents the orientation of the orbital in space. The value of 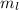 lies between –l to +l. The formula to calculate the value of
lies between –l to +l. The formula to calculate the value of 
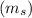 : It represents the direction of the electron spin. Its value can be
: It represents the direction of the electron spin. Its value can be 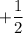 or
or 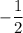 .
.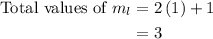
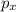 ,
, 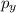 and
and 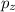 .
.