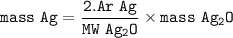Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:20
Aluminum reacts with chlorine gas to form aluminum chloride via the following reaction: 2al(s)+3cl2(g)→2alcl3(s) what is the maximum mass of aluminum chloride that can be formed when reacting 32.0 g of aluminum with 37.0 g of chlorine? express your answer to three significant figures and include the appropriate units.
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2


Chemistry, 22.06.2019 16:00
The chemical equation below shows the reaction of sodium (na) and chlorine (cl) to form sodium chloride (nacl). 2na + cl2 → 2nacl in this equation, which of the following is a reactant? i. sodium ii. chlorine iii. sodium chloride
Answers: 1
You know the right answer?
If one wants 100g of Ag, how much ag2o is needed?...
Questions


Mathematics, 10.07.2019 10:00

Arts, 10.07.2019 10:00

Biology, 10.07.2019 10:00

English, 10.07.2019 10:00

Advanced Placement (AP), 10.07.2019 10:00

Mathematics, 10.07.2019 10:00


Mathematics, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00




Health, 10.07.2019 10:00

History, 10.07.2019 10:00

Biology, 10.07.2019 10:00


Mathematics, 10.07.2019 10:00

History, 10.07.2019 10:00

Mathematics, 10.07.2019 10:00