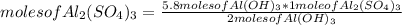Chemistry, 07.02.2021 20:10 salsaanddoritos899
Based on the equation below, how many moles of aluminum sulfate (Al2(SO4)3) will be produced from the reaction of 5.8 moles of aluminum hydroxide (Al(OH)3) and excess sulfuric acid (H2SO4)? 2Al(OH)3 + 3H2SO4 → Al2(SO4)3 + 6H2O

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 14:10
How does chemistry affect our world? a. chemicals makes our world more polluted. b. chemicals keeps us healthy. c. chemicals can or hurt our world. d. chemicals make our world safe to live in.
Answers: 1

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1

Chemistry, 23.06.2019 04:10
An unknown substance has been shown to have weak covalent bonds. which of the following is most likely a property of this substance? a. high ph b. high conductivity c. low melting point d. low flammability
Answers: 3

Chemistry, 23.06.2019 06:40
8. how much enthalpy/heat is transferred when 0.5113gof ammonia (nh3) reacts with excess oxygen according| to the following equation: 4nh3 +502 - 4n0+ 6h20ah = -905.4j
Answers: 1
You know the right answer?
Based on the equation below, how many moles of aluminum sulfate (Al2(SO4)3) will be produced from th...
Questions

Mathematics, 21.04.2020 13:56


Biology, 21.04.2020 13:57

Social Studies, 21.04.2020 13:59





History, 21.04.2020 14:23

Mathematics, 21.04.2020 14:23



Mathematics, 21.04.2020 14:24




Mathematics, 21.04.2020 14:24



Mathematics, 21.04.2020 14:24