
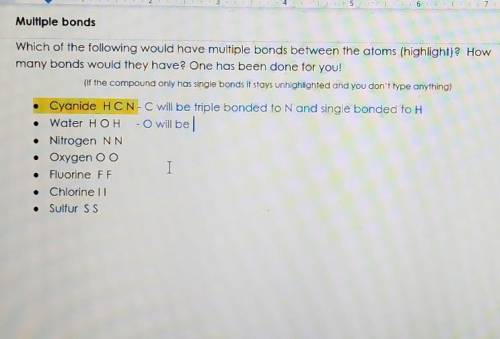
Pls answer quickly, thx. Multiple bonds. Which of the following would have multiple bonds between the atoms (highlight)? How many bonds would they have? One has been done for you! (If the compound only has single bonds It stays unhighlighted and you don't type anything) • Cyanide HCN-C will be triple bonded to N and single bonded to H • Water HOH - will be • Nitrogen NN • Oxygen O O Fluorine FF I Chlorinell • Sulfur SS

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:30
Apiece of metal with a length of 1.42 cm was measured using four different instruments. which of the following measurements is the most accurate?
Answers: 3

Chemistry, 22.06.2019 14:00
The two naturally occurring isotopes of chlorine are 35cl (34.969 amu, 75.77%) and 37cl (36.966 amu, 24.23%). the two naturally occurring isotopes of bromine are 79br (78.918 rm amu, 50.69%) and 81br (80.916 amu, 49.31%). chlorine and bromine combine to form bromine monochloride, brcl. 1. how many peaks will be present in a mass spectrum for brcl? the four combinations of molecule possible given these four isotopes are: 81br37cl, 81br35cl, 79br37cl, and 79br35cl. 2. what are the masses of the four different brcl molecules? express the masses using six significant figures, in decreasing numeric order (highest to lowest), separated by commas.
Answers: 3


Chemistry, 22.06.2019 20:00
Nitrogen dioxide decomposes according to the reaction 2 no2(g) ⇌ 2 no(g) + o2(g) where kp = 4.48 × 10−13 at a certain temperature. if 0.70 atm of no2 is added to a container and allowed to come to equilibrium, what are the equilibrium partial pressures of no(g) and o2(g)
Answers: 2
You know the right answer?
Pls answer quickly, thx.
Multiple bonds. Which of the following would have multiple bonds between t...
Questions

Biology, 19.01.2020 11:31

Biology, 19.01.2020 11:31


Social Studies, 19.01.2020 11:31

Mathematics, 19.01.2020 11:31




Physics, 19.01.2020 11:31

English, 19.01.2020 11:31

Mathematics, 19.01.2020 11:31



Mathematics, 19.01.2020 11:31

Biology, 19.01.2020 11:31

History, 19.01.2020 11:31


Mathematics, 19.01.2020 11:31

Mathematics, 19.01.2020 11:31



